A Device That Costs Pennies Could Prevent Millions Of Child Drug Overdoses: The Best Liquid Medicine Flow Restrictors On The Market [LIST]

A potential drug overdose could occur in the few moments a parent glances away from their child as the curious youngster reaches over the counter to grab a medicine bottle for a taste of the syrupy contents. Pediatric liquid medications, such as cough and cold medicines and acetaminophen, left unattended can lead to the swallowing of potentially toxic doses that place tens of thousands of kids in the emergency room each year. Dr. Daniel Budnitz, a scientist at the Centers for Disease Control and Prevention’s (CDC) Medication Safety Program, believes the implementation of a flow restrictor — a safety valve that fits into the neck of a medicine bottle and slows the release of fluid — in all pediatric versions medicines could significantly reduce the number of pediatric ER visits for drug accidents.
The CDC reports each year, approximately 53,000 children less than 5-years-old, go to ERs for unsupervised ingestions of over-the-counter cough and cold medicines. In October, leading manufacturers of children’s cough and cold medicines addressed safety concerns in regards to the labeling of their products. Previously, cough and cold medicine labels would state these products should not be given to children under age 2, but now the age has been raised to no younger than 4 after an unprecedented number of drug-related pediatric ER visits.
In 2011, as part of a pledge, drug makers added flow restrictors to infants’ and children’s products with acetaminophen, according to ProPublica, an independent, nonprofit newsroom. The implementation of the small plastic device reduced the number of kids’ ER visits for drug overdoses involving pediatric or adult formulas of acetaminophen by 25 percent. However, industry officials remain weary on the use of flow restrictors until they are presented with better data that solidifies these devices lessen kids’ risk from drug overdoses.
The safety initiatives by manufacturers have led to a reduction in kids’ ER visits for drug accidents, but some pediatricians, like Dr. G. Randall Bond, believe more needs to be done. "If flow restrictors work, they should be placed on all liquid products," he told ProPublica. “We need a technological change to get us to the next level of safety.”
Currently, there are no standards for the design of flow restrictors and no public data present on how effectively the different types of these small devices work. While manufacturers remain on the fence about including flow restrictors in children’s medications, Consumer Reports tested flow restrictors on more than 30 liquid infant acetaminophen bottles found on drugstore shelves across the United States. The company has shared its results with ProPublica, who also has its ongoing investigation into acetaminophen safety.
Consumer Reports first measured the physical abilities of a 4-and-a-half-year-old boy — the age range that child-resistant packing is commonly designed to protect — to see the ways a child would be most likely to remove the medicine from the bottle by squeezing, shaking, or sucking on it. The products used in the tests did not identify on the packaging what kind of flow restrictor they used, but the scientists were able to differentiate these restrictors into two basic categories.
The products contained either open flow restrictors of closed flow restrictors. According to Consumer Reports, open flow restrictors contain a plastic disc with a small hole in the center to prevent an unattended child from quickly emptying the contents of the bottle. Similar to its counterpart, closed flow restrictors contain a plastic disc but include an elastic valve in the small hole that opens when the tip of the syringe is placed in the bottle and then closes automatically when it's removed.
In general, flow restrictors help prevent an unattended child from drug accidents, but the closed versions are found to be more effective in greatly reducing or completely eliminating the quantity of liquid a child could dispense from the medicine bottle. PediaCare, an acetaminophen children’s medicine brand, was found to be the most effective with a closed flow restrictor compared to Tylenol products, which have an open flow restrictor.
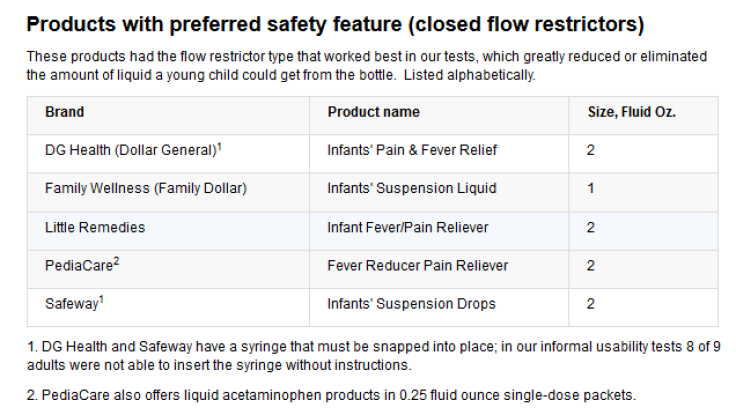
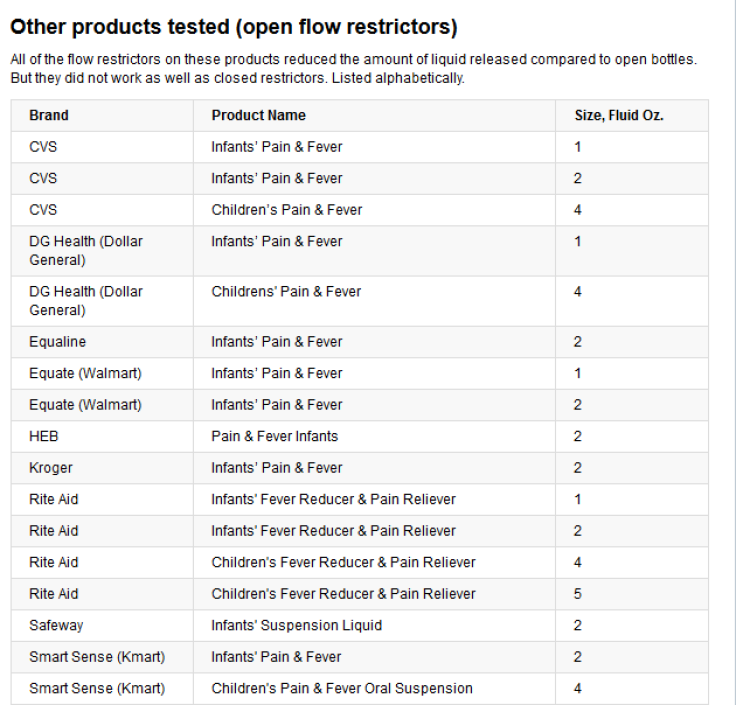
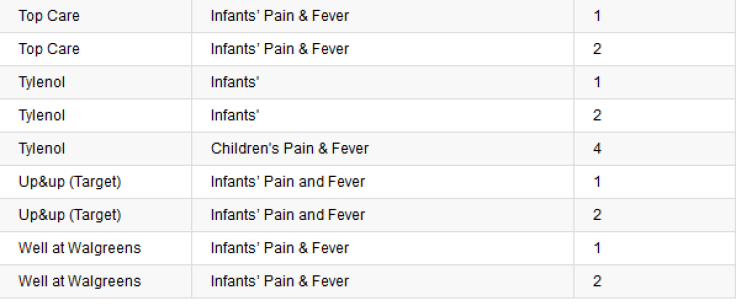
McNeil Consumer Healthcare, the Johnson & Johnson unit that makes Tylenol, was contacted by Consumer Reports and ProPublica with questions about the test results, but the company failed to directly address why it chooses a less efficient flow restrictor. “McNeil continues to evaluate feedback and data on the effectiveness of flow restrictors and, based on the results of this data, will determine whether to extend the use of flow restrictors beyond Infants’ and Children’s Tylenol,” the company said in a statement.
Packaging experts remain skeptical about Tylenol and other companies who claim to extend the use of flow restrictors to all their products. Clay Robinson, a packaging expert whose company tests safety devices for drug makers, told ProPublica, “If it’s going to add cost, it’ll be hard to get industry interested.”
Child safety packaging continues to remain a high concern for pediatricians and parents. Until children’s cough and cold medicine and acetaminophen makers include flow restrictors in all their products, parents are advised follow the CDC’s basic tips:
- Pick a place your children cannot reach to keep medicines
- Put every medicine away every time you use it
- Always relock the safety cap every time you open it
- Teach your children about medicine safety
- Tell guests about medicine safety
- Program the Poison Help number into your phone: 1-800-222-1222



























