Mini-Kidney Grown From Stem Cells Holds Promise As Diagnostic Tool, Renal Function Booster
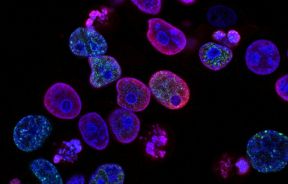
It’s barely the width of a pin head, but it’s a kidney all right. Australian researchers from the University of Queensland have developed the mini-organ, known as an organoid, from a collection of self-assembling stem cells, which they predict may have integral function in diagnosing renal diseases, and hopefully, one day treating those diseases.
Published in the journal Nature Cell Biology, the new study adds to a growing collection of organs that were built via stem cells. A first of its kind, the breakthrough development is slated to usher in compounding research into how organs can be built, both as experimental tools where scientists can see firsthand how diseases form and as replacement organs for patients whose current systems are failing. While the prospect of implanting a full-grown stem cell kidney is still a decade away, according to the team, the steps to get there hold great promise for future investigations.
Stem cell spleens and lungs no doubt serve a medically scientific purpose, but in terms of fulfilling a demand, kidneys have few rivals. In the U.S. alone, chronic kidney disease affects roughly 20 million people — or 10 percent of the population over 20 years old — and its prevalence only increases with age, according to the Centers for Disease Control and Prevention (CDC). End Stage Renal Disease (ESRD), also known as kidney failure, is typically the end result of a progressive and irreversible condition. Patients with ESRD must undergo regular dialysis as they wait for a kidney transplant, if one ever comes.
Lead researcher of the new study, Dr. Melissa Little, concedes her team’s mini-kidney is still a decade or so away from a working full-size version, one that can reliably be transplanted into a patient. In the meantime, the development is poised to save millions of dollars in renal disease research.
“It costs half a billion dollars to get any drug to market, and that’s largely because most drugs fail and fail late,” Dr. Little, of UQ’s Institute for Molecular Bioscience, told The Australian. “Three organs are often damaged by drugs: the liver, heart, and kidney. If you could work out earlier which drugs were toxic to the kidney, you wouldn’t spend half a billion dollars.”
In this, the mini-kidney could potentially serve a range of functions. Scientists hope to bundle a cluster of mini-kidneys together someday and essentially construct a full-size kidney in the aggregate, which could then be given to a patient. They also hope to begin producing nerve cells within the organs, as prior experimenters have made “some advances,” Little says, in crafting heart and blood cells and components of the eye and brain. What remains a challenge, however, are the larger organs that work more in isolation from one another.
“There are laboratories all over the world trying lung, liver, pancreas and many different organs,” Dr. Little explained. “This is a very exciting field that has a lot of possibilities long term.”
In order to construct the mini-kidney, scientists had to comb through countless genes and determine which ones responded in the desired self-organizing pattern when switched “on” or “off.” Professor Brandon Wainwright, also from UQ, told the Telegraph that the process was akin to “a scientific approach to cooking,” as the researchers relied on a trial-and-error method to tease out the appropriate genes.
After successfully nailing down these genes, after a period of some years, Wainwright said, the team manipulated the skin cells into embryonic stem cells, which they then used to form the complex human structure.
"It is an amazing process — it is like a Lego building that puts itself together,” Wainwright told the Telegraph. You can eventually coax these stem cells through a journey — they [the cells] go through various stages and then think about being a kidney cell and eventually pop together to form a little piece of kidney."
Source: Takasato M, Becroft P, Vanslambrouck J. Directing human embryonic stem cell differentiation towards a renal lineage generates a self-organizing kidney. Nature Cell Biology. 2013.