Origins of “Timezyme” Discovered: Melatonin-Producing Enzyme Crucial For Regulating Your Internal Clock
Just as the mountain ranges and canyons of earth are linked to specific geologic events occurring in historic periods, so too do structures, hormones, and biochemical processes of our bodies trail backwards to a point of origin. Now, an international team of scientists has traced the likely genesis of a crucial enzyme found within our bodies to roughly 500 million years ago. Led by researchers from the National Institutes of Health (NIH), the scientific team hypothesizes that an enzyme producing melatonin, which plays a role in regulating the body’s internal clock, likely evolved when vertebrates (animals with spinal columns) diverged from their nonvertebrate ancestors.
Melatonin and Circadian Rhythms
Melatonin, which is manufactured in the brain’s pineal gland, is a key hormone that regulates the body’s circadian rhythm — an internal 24-hour “clock” that helps us fall asleep and wake up, and even impacts our aging. Melatonin, which is produced from the hormone serotonin, is actually the end product of a sequence of chemical reactions. The penultimate step in this multi-step process consists of attaching a small molecule (the acetyl group) to the nearly-complete melatonin molecule. This acetylation step is performed by an enzyme called arylalkylamine N-acetyltransferase, or AANAT.
AANAT is often referred to as the "timezyme" because of its role in producing melatonin, the regulator of your body’s internal time clock. Melatonin is a "hormone of the night": In vertebrates, including humans, circulating levels at night are markedly higher than levels during the day. This increase is driven by precisely regulated increases in acetylation by AANAT in the pineal gland. Yet small amounts of melatonin are also found in the retina of the eye, and one form of AANAT, or timezyme, appears to detoxify the eye when potentially hazardous compounds enter those delicate tissues. This form of timezyme, however, is found only in non-vertebrates.
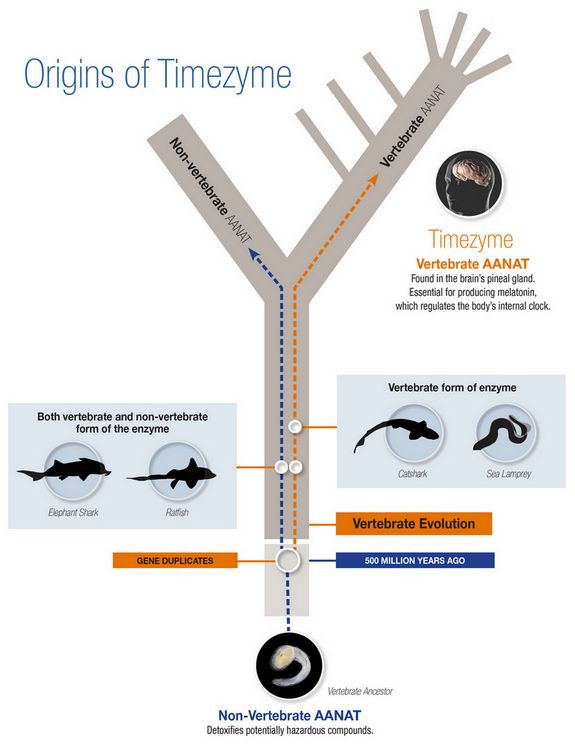
Because of this, senior author David Klein, Ph.D., NIH, and his team of researchers theorized that a second copy of the gene for producing AANAT appeared about 500 million years ago, when the original gene was duplicated. Gene duplication typically results from any of a number of genetic mishaps during cell division. Instead of one copy of a gene resulting from the process, an additional copy results, so that there are two versions of a gene where only one existed previously.
As vertebrate animals evolved, the researchers' new hypothesis suggests, the second copy of the AANAT gene similarly evolved and eventually specialized in producing melatonin. Their theory also states that the original copy of the AANAT gene later disappeared, and its function was taken over by other genes. Before the current study, though, the researchers lacked proof of their elaborate theory, particularly in regard to the question of how the vertebrate form of the enzyme originated because it did not appear to exist in non-vertebrates and had been found only in bony fishes, reptiles, birds, and mammals — all of which lacked the non-vertebrate form. After Steven L. Coon, Ph.D., NIH and study co-author, discovered genes for the nonvertebrate and vertebrate forms of AANAT in genomic sequences from the elephant shark, considered to be a living representative of early vertebrates, the new hypothesis gained ground.
Two Timezymes?
After analyzing DNA from various sea creatures, the researchers found supporting evidence for their theory of the origin of melatonin and timezyme. The researchers discovered, for instance, the elephant shark and the ratfish — two animals thought to be similar to early vertebrates — produce both the non-vertebrate and vertebrate forms of AANAT. In comparison, two other animals thought to have originated later in vertebrate evolution— the catshark and the sea lamprey — had only the vertebrate AANAT gene.
“Nonvertebrate AANAT appears to detoxify a broad range of potentially toxic chemicals,” Klein said in a press release. “In contrast, vertebrate AANAT is highly specialized for adding an acetyl group to melatonin. The two are as different from each another as a Ferrari is from a Model-T Ford, considering the speed of the reaction and how fast it can be turned on and off.”
The team’s findings, then, provide support for a working theory that the timezyme originated to remove toxic compounds from the eye and then gradually became a switch for controlling the body’s 24-hour cycle. The scientists believe a deeper understanding of the enzyme’s function, both before and after the evolutionary divergence, could contribute to an understanding of melatonin-related conditions, including seasonal affective disorder, jet lag, and even vision disorders.
Source: Klein DC, Falcon J, Coon SL, et al. Drastic neofunctionalization associated with evolution of the timezyme AANAT 500 Mya. PNAS. 2013.
Published by Medicaldaily.com



























