Siege On Plasmodium Falciparum: Targeting Protein In Malarial Parasite May Block Inflow Of Nourishment, Destroying Parasite
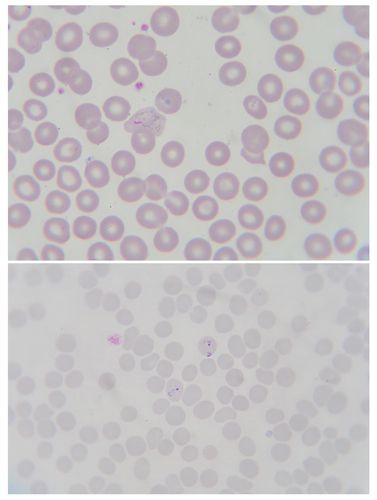
Plasmodium falciparum, the parasite which causes malaria, evades the host immune system by a variety of mechanisms and causes persistent, recurrent infections. But scientists from Washington University School of Medicine in St. Louis have found a way to use the parasite’s attacking machinery against it and confine it in its own membrane, where it will eventually perish due to lack of nourishment. The research appears in Nature.
P. falciparum is the deadliest malarial parasite and is known to cause around 2.7 million deaths annually. It is endemic to several parts of the world, but 90 percent of the deaths occur in sub-Saharan Africa. The parasite is transferred to a human body from the bite of an infected mosquito.
As it directly enters the blood stream, it invades the red blood cells. It then makes use of the cell's membrane and builds a protective barrier around it. Then, for the next 48 hours during its asexual reproduction stage, the parasite causes massive changes in the host cell, making it permeable so that nutrients can move in and waste matter out. These changes are brought about by hundreds of proteins, which are exported by the parasite from its membrane into the hosts. This new research suggests the proteins pass through a single pore in the parasite's protective barrier into the red blood cell. When this pore was blocked in cell cultures, the parasite stopped growing and died.
"The malaria parasite secretes hundreds of diverse proteins to seize control of red blood cells," said first author Josh R. Beck in a press release. "We've been searching for a single step that all those various proteins have to take to be secreted, and this looks like just such a bottleneck."
The importance of this pore in the parasite’s survival has also been highlighted in research published in Nature by researchers at the Burnet Institute and Deakin University in Australia. It is believed that disrupting passage through this pore leaves the parasite fatally imprisoned, unable to steal resources from the red blood cell or dispose of its wastes.
Senior author Daniel Goldberg is studying the effects of the parasite on red blood cells. The parasite destroys red blood cells, causing anemia. It also latches on to organ membranes like lungs, kidneys, and even the brain, leading to severe complications. As more and more resistant strains of the pathogen emerge, conventional drugs seem to lose their effectiveness, prompting scientists to discover new drug targets.
In this research, scientists looked into the possible role of protein heat shock protein 101 (HSP101), so-called because they are expressed in high quantities in response to heat and other stresses. The proteins have multiple functions, including guiding the folding and unfolding of other proteins.
Previous studies have shown that the protein may be activating protein secretion pathways, so when the researchers disabled HSP101 in cell cultures, they hoped to block discharge of some malarial proteins. But to their surprise, it blocked all.
"We think this is a very promising target for drug development," Goldberg said. "We're a long way from getting a new drug, but in the short term we may look at screening a variety of compounds to see if they have the potential to block HSP101."
HSP101 may be involved in the secretion of malarial proteins into the host red blood cells through the pore. In order to this, scientists believe that it unfolds the proteins into a linear form so that they can easily pass through the narrow pore. HSP101 may also initiate a biochemical process that gives the proteins the necessary momentum to get through the pore.
While Beck and his team targeted the HSP101 protein, the Australian researchers neutralized the parasite in a similar fashion by disabling another protein thought to be involved in the passage of proteins through this pore.
"That suggests there are multiple components of the process that we may be able to target with drugs," he said. "In addition, many of the proteins involved in secretion are unlike any human proteins, which means we may be able to disable them without adversely affecting important human proteins."
Source: Beck J, Muralidharan V, Oksman A, Goldberg D, HSP101/PTEX mediates export of diverse malaria effector proteins into the host erthyrocyte, Nature. 2014.
Published by Medicaldaily.com