Targeting Alzheimer’s Disease: Protein 'Chaperone' Contributes To Build-Up Of Brain Plaque
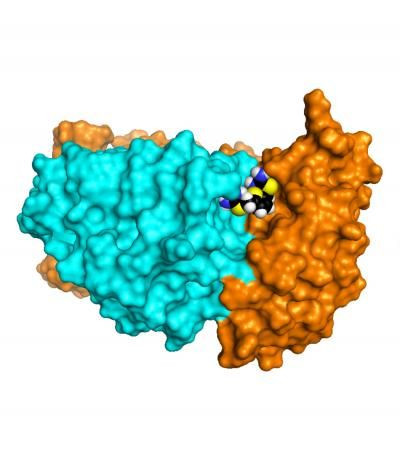
In his past research, Dr. Scott Small, director of the Alzheimer's Disease Research Center at Columbia University Medical Center (CUMC), showed that retromer protein complex is deficient in the brains of patients with Alzheimer's disease. Now, his latest research conducted with researchers from Weill Cornell Medical College and Brandeis University has identified a new class of compounds that can significantly increase retromer levels while decreasing amyloid-beta levels in cultured neurons. These pharmacologic "chaperones" may represent an entirely new method for treating Alzheimer's disease. "This approach may prove to be safer and more effective than conventional treatments for neurologic disease, which typically target single proteins," said Dr. Small, senior author of a paper on the subject appearing in Nature Chemical Biology.
Brain Plaque
A key marker of Alzheimer’s disease is the plaque formed when beta-amyloid protein pieces clump together. Most people develop some plaque building up in the spaces between nerve cells as they age. When too much plaque accumulates, it contributes to the development of Alzheimer's, a degenerative brain disease, which causes problems with memory, thinking, and behavior and progresses over time until symptoms become so severe a person cannot function. There is no cure for Alzheimer's, though. Many scientists are hoping to develop a drug that would slow the progress of the disease or ease its symptoms.
To that end, Small and his colleagues have observed how retromer abnormalities occur in the brains of people with Alzheimer's with similar aberrations linked to Parkinson's disease. Understanding this, Small and his team of researchers considered the complex of proteins might become a worthy drug target. The retromer protein complex seemed to steer amyloid precursor protein (APP) away from a region of the brain cell where normally APP is reduced (if not destroyed), and when this happens, it created a byproduct of beta-amyloid. In short, when this protein complex functioned incorrectly, it appeared to contribute to the development of plaque in the brain. "Our findings identify a novel class of pharmacologic agents that are designed to treat neurologic disease by targeting a defect in cell biology, rather than a defect in molecular biology," said Small in a press release.
Having identified a target, the team went to work looking for a way to stabilize retromer and bolster its function. Examining past scientific literature, they saw how other researchers had mapped out the retromer's three-dimensional structure. "Our challenge was to find small molecules—or pharmacologic chaperones—that could bind to retromer's weak point and stabilize the whole protein complex," said Dr. Dagmar Ringe of Brandeis University and co-author of the paper.
Using a computerized virtual, or in silico, screening of known chemical compounds, the team simulated how the compounds might dock with the retromer protein complex. The screening identified 100 potential retromer-stabilizing candidates, 24 of which showed particular promise. Of those, one compound, called R55, was found to significantly increase the stability of retromer when the complex was subjected to heat stress.
Next, the team of researchers examined how R55 affected neurons in the hippocampus, a key brain structure that is affected by Alzheimer’s as it is involved in memory and learning. Through experimentation, the researchers found the compound significantly increased retromer levels and decreased amyloid-beta levels in cultured neurons taken from healthy mice and from a mouse model of Alzheimer's. "One concern was that this compound would be toxic," said Dr. Diego Berman, assistant professor of clinical pathology and cell biology at CUMC and a lead author. "But R55 was found to be relatively non-toxic in mouse neurons in cell culture."
Currently, the researchers are testing the clinical effects of R55 in an actual mouse model. "The odds that this particular compound will pan out are low, but the paper provides a proof of principle for the efficacy of retromer pharmacologic chaperones," said Dr. Gregory A. Petsko, director of the Helen and Robert Appel Alzheimer's Disease Research Institute at Weill Cornell Medical College. "While we're testing R55, we will be developing chemical analogs in the hope of finding compounds that are more effective."
Source: Small S, Mecozzi VJ, Simoes S, et al. Pharmacological chaperones stabilize retromer to limit APP processing. Nature Chemical Biology. 2014.
Published by Medicaldaily.com