What Is The Top-Secret Ebola Serum That May Have Saved The Lives Of Two US Patients?
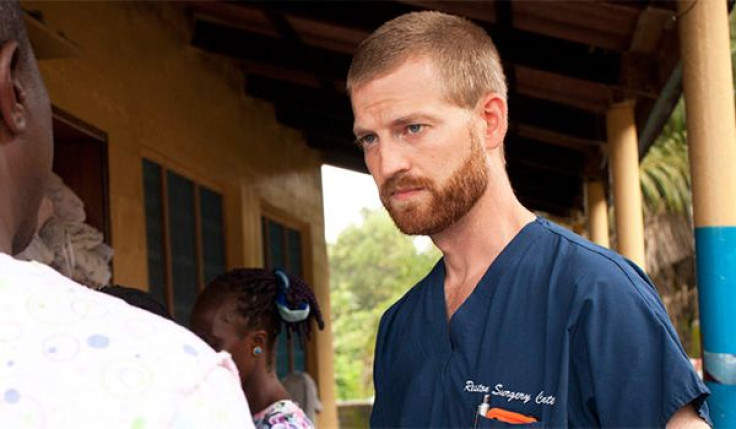
If you’ve been following the current Ebola outbreak in West Africa, you may be familiar with the experimental "top-secret serum" administered to the first two American patients, Dr. Kent Brantly and his colleague, Nancy Writebol. As more reports come in of Brantly’s "miraculous" Ebola recovery and Writebol’s steady improvements, the world wants to know what this mysterious serum is and if it's the cure we’ve all been waiting for.
“Three vials of the top-secret serum were flown into Liberia last week in a desperate effort to save the lives of American missionary workers, Brantly and Writebol,” Dr. Sanjay Gupta, CNN chief medical correspondent explained in his report. The effects of the serum on Brantly were noticed within hours of its administration. The Atlantic reported that one of the doctors working at the Emory University Hospital, where Brantly is being treated, described the serum’s effects as "miraculous," and Gupta reported that Brantly has now made a “near complete recovery.” Writebol’s condition has also improved following the treatment, although not as drastically as Brantly.
The mystery serum whose abilities have left both scientists and laymen at a loss for words is known as ZMapp. Here’s what we know about the drug so far: Brantly and Writebol are the first humans to ever use the experimental serum. It has previously only been tested on eight monkeys. In the animal trials, all monkeys who were administered the serum within 48 hours of contracting Ebola survived. Those who were given the drug outside of the two-day time window did not recover; however, both Brantly and Writebol were administered the drug outside of the 48-hour window and seem to be doing well.
Scientifically speaking, there is nothing groundbreaking about ZMapp. It’s a monoclonal antibody, a common disease-tackling approach that has been used by scientists for some time now. Monoclonal antibodies come about when scientists infect an animal with a disease, collect the antibodies the animals create to fight the disease, and then administer those antibodies to a human.
Unfortunately, as Business Insider reported, a cure for Ebola may still be quite far off and, at this point, it may be more likely to contain the virus than cure it. Still, the U.S. military’s Defense Threat Reduction Agency made a promising move when they recently approved additional funding for Mapp Biopharmaceutical, the creators of ZMapp. Although most drugs require years of testing before being approved, Gupta believes the FDA may allow the drug to forgo the usual trial period through the use of the compassionate use exemption.
Why it’s taken the death of 887 Africans before ZMapp was offered is a controversial issue. Experts explain that in the past lack of enthusiasm in developments for Ebola drugs have been due to the previous rareness of the virus combined with concern that sales of such a drug would not produce enough revenue for its pharmaceutical companies.



























