Asfotase Alfa Granted Breakthrough Status By FDA For Treatment of Childhood Bone-Softening Disease, Hypophosphatasia
Alexion Pharmaceuticals, a small pharmaceutical company out of Switzerland which focuses on the development of drugs for rare diseases, has announced that the U.S. Food and Drug Administration (FDA) has given it a special designation for one of its new investigational drugs.
The Breakthrough Therapy designation was given to Asfotase Alfa, which is meant to treat hypophosphatasia (HPP), a disease in which the organs slowly self-destruct and bones also slowly weaken and deform. The breakthrough status was granted because there is currently no treatment for the life threatening disease and the new drug offers a leap forward for treatment in children with the disease.
"The FDA's Breakthrough Therapy designation for perinatal-, infantile- and juvenile-onset HPP recognizes the severe, debilitating and life-threatening nature of the disease, the clear unmet medical need of patients, and the clinical evidence collected to date on asfotase alfa," said Martin Mackay, Ph.D., Executive Vice President, Global Head of R&D at Alexion. "Asfotase alfa is a highly innovative therapeutic candidate with the potential to transform the lives of patients with HPP who currently have no treatment options and often receive only palliative care for this life-threatening disease."
The drug is targeted at children who have just been born, in infants, and in and young children who were diagnosed with the disease before they become 18 years of age.
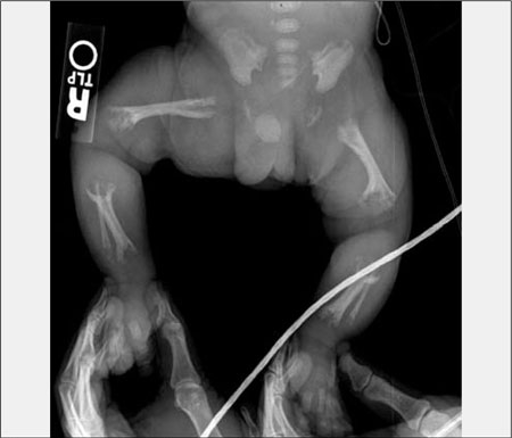
The life-threatening disease is genetic in nature — it is caused by a mutation in a gene called tissue non-specific alkaline phosphatase (TNSALP). The result of the mutation is that the body cannot properly regulate calcium and phosphate, two minerals necessary for bone development, proper organ function, muscle function, kidney function, and respiratory function. Bone is made up of mineralized calcium and phosphate and the disease was named for the obvious skeletal deformities it can cause.
The disease can be detected by ultrasound analysis of the fetus before birth and genetic tests of amniotic fluid. The first symptoms appear before six months of age and children display skeletal abnormalities and breathing problems leading to a dismal 50 percent mortality rate. The disease is classified as an "orphan disease," because it is so rare and there are currently no approved treatment options. The incidence of the disease is estimated to be one in every 100,000 live births and the genetic cause is inherited from parents who are carriers most of the time.
The Alexion drug, Asfotase Alfa, is an enzyme replacement therapy that increases the amount of disease-causing mutated enzyme with a version that works properly. The drug is capable of treating, preventing, and reversing the complications of the genetic disease. For this reason, the FDA has granted the Breakthrough Status, which will expedite the approval process for the drug and allow patients to be treated with it quicker.
To watch a video of a parent's and child's struggle with the disease and how Alexion has helped click here.
Published by Medicaldaily.com