Deadly Brain Cancer Glioblastoma Multiforme Could Soon Be Treatable, Thanks To Discovery Of Key Growth Instigator

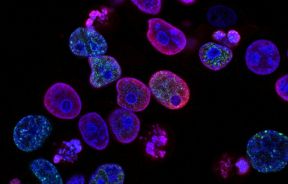
Any kind of cancer is terrifying, but brain cancer may be the scariest of the bunch. Among these cases, however, some stand out as particularly deadly — glioblastoma multiforme is one such form. A person diagnosed with this type of brain tumor is typically given about 15 months to live, even with the best possible care. A team of researchers at the Salk Institute have taken on the deadly cancer, though, and succeeded in finding a key reason behind why these particular tumor cells proliferate so quickly.
"This is a disease for which there has been practically no improvement in treatment outcome for years," said Inder Verma, professor at the Salk Institute’s Laboratory of Genetics and senior author of the paper, in a press release. "It is clear that even if a surgeon removes 99.99 percent of a glioblastoma multiforme tumor, what is left behind will come back and grow into more tumor." To get more familiar with how the tumor spreads, Verma’s team examined a transcription factor (a protein that binds to DNA and controls gene expressions for a particular set of genes) called kBor NF-kB.
There are several known factors that can trigger NF-kB activity in a cell, like immune proteins, DNA damage, and ultraviolet and ionizing radiation. The researchers took several tests to see how overzealous NF-kB activity influenced cancer cells to multiply, and how halting the transcription factor might slow cancer growth and increase survival time.
"Our experiments confirmed that NF-kB is required for the cancer cell to proliferate," said Dinorah Friedmann-Morvinski, a researcher at the department of biochemistry and molecular biology at Tel Aviv University in Israel and first author of the paper. "But now we have finally found a way to ameliorate the tumor to increase lifespan."
Verma and his colleagues used genetic tools to manipulate the cells of a mouse model of glioblastoma multiforme, trying different ways to shut down NF-kB activity; first, they tried to up the presence of a protein called IlBaM, which inhibits the activity of NF-kB. Next, they eliminated an enzyme that boosts NF-kB activity. With less NF-kB action, tumor growth slowed down, and the mice lived significantly longer than mice whose NF-kB activity was left alone. These genetic treatments showed great results, but unfortunately, they aren't exactly feasible in humans.
Verma explained they then tried to find a way to manipulate the system using pharmacology rather than genetics. It has been suspected that one reason why this type of tumor comes back so quickly after surgery is what's called the tumor microenvironment. Basically, a tumor changes its surrounding environment to make it easier for cancer cells to grow and thrive. Knowing this, the team switched their focus to an approach that would also change the tumor microenvironment.
They fed the mice a peptide called NBD, which is known to block NF-kB activity in the scenario where it is triggered by cytokines. The peptide easily travels across the central nervous system and has the ability to penetrate glioblastoma tumor cells. NBD's capabilities paid off — the mice treated with it doubled their typical survival time compared to mice that did not receive the peptide treatment.
"We could increase survival time from one month without treatment to three months with treatment," Verna said. "That's a profound increase in life expectancy, especially considering a mouse only lives for two years."
There is a downside, though: the peptide treatment eventually causes toxicity, usually in the liver. So the researchers went back again and tried to explore a new tactic. NF-kB can be tricky to curb because it has many important roles, including regulation of immunity, inflammation, and other functions.
"The ultimate goal is to block NF-kB, but because it turns on many genes — at least 100 — our aim became finding the handful of genes that directly affect tumor grown,” Verma said. “Then we can be more selective in treatment."
The team did track one gene, Timp1, influenced by NF-kB that has previously been implicated in lung cancer; targeting the gene in treatment slowed tumor growth and increased survival time.
"In the future we want to focus on ways to reduce the toxicity of anti-NF-kB drugs," Friedmann-Morvinski said. "We may do this by specifically targeting these drugs to the tumor, or by identifying downstream targets of the NF-kB pathway, like Timp1, that also prolong survival."
Source: Verma I, Friedmann-Morvinski D, Narasimamurthy R, Xia y, Myskiw C, Soda Y. Targeting NF-kB in gliblastoma: a therapeutic approach. Science Advances. 2016.