Dengue Vaccine Benefits And Risks: New Study Notes Dangerous Regions
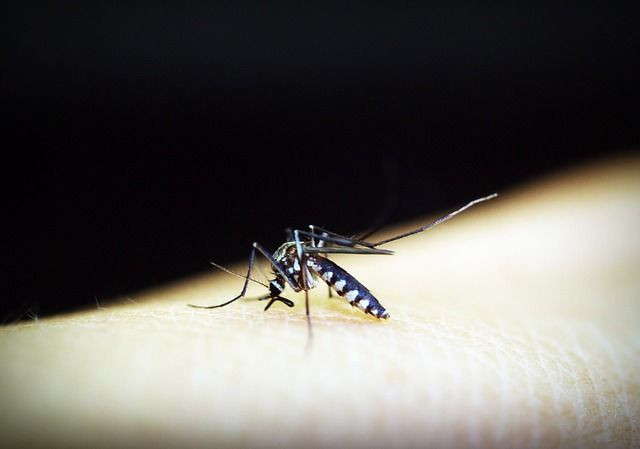
A new study published Thursday in the journal Science suggests the world’s first approved dengue vaccine may harm people if improperly distributed.
While Dengvaxia can prevent people who have already survived dengue from coming down with a second devastating infection, it can also make people who weren’t previously exposed to dengue more vulnerable to a later infection down the road, so it only makes sense to vaccinate people in areas with a high risk of transmission. Although Dengvaxia was created to immunize someone from all four strains, it’s only moderately effective, so the chances of someone not being fully protected are more than likely.
Currently, the three-dose vaccine is approved for use in six countries, and it has been endorsed by the World Health Organization (WHO), although only for areas of the world with high dengue transmission. It's estimated that 50 to 100 million people are infected by dengue annually in around 120 countries, with nearly half the world's population at risk, according to the WHO.
For the study, a collaboration of researchers took another look at data from earlier trials of Dengvaxia, created by the French-based pharmaceutical company Sanofi (and its vaccine branch, Sanofi Pasteur). The original trials enrolled more than 30,000 participants across ten different countries, and the researchers also looked at more recent studies that examined the long-term health outcomes of these participants. They then created a computer model that predicted how effectively the vaccine would work in countries with varying levels of dengue transmission. Though the vaccine would work as advertised in areas of high transmission, reducing the risk of severe illness and hospitalization by as much as 30 percent, it could do the opposite in areas of lower transmission, the researchers calculated.
"In vaccines you hope for more than 30 percent success, but it's the only vaccine available right now to slow dengue," said lead author Dr. Isabel Rodriguez-Barraquer, a research associate at the Johns Hopkins Bloomberg School of Public Health Bloomberg School, in a statement. "If this vaccine is used correctly, many people could be spared illness and hospitalization from dengue. But we should make sure we only use it in places where our data suggest it will do more good than harm."
The differing success rates are due to a particular quirk of the dengue virus, which is transmitted by female Aedes mosquitoes. There are four major strains of the virus, and surviving an infection only protects someone from whichever particular strain they were infected by. While dengue is typically not serious the first time someone gets it (only one-fourth of carriers experience any symptoms, which often resemble the flu), the risk of severe illness, such as a hemorrhagic fever that causes internal bleeding and shock, becomes increasingly high when that person is infected with a different strain. The vaccine also isn’t recommended for children under the age of nine, since they’re less likely to have survived an earlier infection.
Far from demonizing Dengvaxia, the researchers hope their findings only highlight the nuanced reality of modern-day medicine, namely that there’s no such thing as a perfect drug; a crucial reminder in light of ongoing research underway to develop vaccines for other mosquito-borne viruses such as Zika and chikungunya.
"Having a vaccine is a significant step forward for dengue control," Rodríguez-Barraquer said. "However, this vaccine is a prime example of having to seriously weigh the risks and benefits."
Source: Ferguson N, Rodriguez-Barraquer I, Dorigatti I, et al. Benefits and risks of the Sanofi-Pasteur dengue vaccine: Modeling optimal deployment. Science. 2016.
Published by Medicaldaily.com



























