Vitality
How to Live Better, Longer

Healthy Evening Routine: Sleep Wind-Down Habits to Balance Your Brain, Gut, and Hormones Naturally
Discover a healthy evening routine that supports deep sleep, balances hormones, and strengthens the gut-brain connection through mindful bedtime habits and natural sleep wind-down practices. 
High‑Protein Diets: How Much Do You Actually Need for Weight and Muscle
Discover how a high-protein diet supports muscle gain and weight loss. Learn your ideal daily protein needs and grams of protein per day for optimal results. 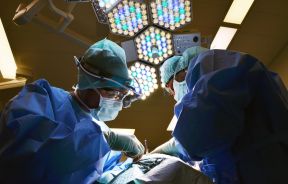
Fat Transfer vs Implants: Natural Body Contouring and Fat Grafting Surgery for Breast and Buttock Augmentation
Learn the differences between fat transfer vs implants, natural body contouring, and fat grafting surgery for breast and buttock augmentation to choose the best cosmetic enhancement options. 
7 Protein Myths Debunked: The Truth About Protein Intake, Muscle & Kidney Health
7 protein myths debunked with science-backed protein intake facts, kidney health insights, and realistic guidance on high protein diets.
Mental Health

Burnout or Depression? Key Differences and How Experts Diagnose Emotional Exhaustion
Burnout vs depression explained: learn emotional exhaustion signs, key differences, and how experts make a mental health diagnosis accurately. 
Teen Mental Health in the Digital Age: Surprising Social Media Effects and Insights from Adolescent Psychology
Explore how social media effects shape teen mental health, highlighting key findings from recent adolescent psychology research plus practical insights for parents, educators, and caregivers. 
Depression Warning Signs in Adults: What Doctors Want You to Know About Hidden Symptoms
Learn how to recognize early depression symptoms in adults. Discover key depression warning signs doctors highlight to protect and improve adult mental health. 
Anxiety Disorders: Key Mental Health Symptoms, Common Triggers, and Proven Treatments
Discover what anxiety disorders are, their mental health symptoms, common triggers, and the most effective anxiety treatment options to help manage stress and improve well-being.
Innovation

Smart Thermostats and Health: How Bedroom Temperature Affects Breathing
Discover how smart thermostats and health are connected by optimizing bedroom temperature to improve sleep quality, support easier breathing, and create a more comfortable, restorative sleep environment. 
IVF Treatment in 2026: Step by Step Process, Success Rates, Costs, and Alternatives
Learn the IVF process step by step, success rates by age 35-40, 2026 treatment costs, embryo transfer, and fertility alternatives like IUI and egg freezing. 
Service Robots Revolutionize Healthcare, Retail, and Hospitality With Smart Automation
Service robots are reshaping healthcare, retail, and hospitality by improving efficiency, safety, and daily operations through smart automation. 
CES 2026 Breakthroughs in Medical & Wellness Technology
Explore CES 2026 breakthroughs in medical and wellness technology, from AI diagnostics to next-gen wearables, highlighting benefits, risks, and the future of connected health innovation.
Healthy Living

Still Hungry After a Meal? The Science of Food Cravings and Why We Crave Food When Full
Why do food cravings appear when you are already full? Learn the science of food cravings after eating and what drives those extra snack urges. 
Strength Training Over 40: Joint-Friendly Workouts and Bone-Strengthening Exercises for a Faster Metabolism
Boost strength and vitality after 40 with simple resistance training. Discover joint-friendly workouts, bone-strengthening exercises, and metabolism-boosting routines for lasting health and energy. 
Immune-Boosting Foods: What Really Works According to Evidence-Based Nutrition (and What's Just Marketing Hype)
Discover the truth about immune-boosting foods with evidence-based nutrition. Learn to spot immune health myths, supplement hype, and food vs marketing claims effectively. 
How Immune Resistance Works: Signs of a Weak Immune System and How to Strengthen Your Body's Defenses
Immune resistance affects how your body fights infections. Learn signs of a weakened immune system and ways to strengthen immune system resistance naturally.
Covid - 19

SKNV is the First Pharmacy for Sensitive Skin
It’s reported that up to 70% of the population have sensitive skin. More alarming, allergies and skin sensitivities are known to affect millions of patients taking prescriptions every year. 
No Time to Exercise? How to Lose Weight and Get in the Best Shape You've Ever Been
Beyond Body can help you get in the best shape even without exercise. Here's how. 
How Red Borneo Kratom Boosts Your Productivity
Red Borneo Kratom can help increase your productivity. 
Nolah Mattress Review: Is It a Good Buy?
Read our full Nolah Mattress review on their AirFoam mattress here.







