Immune System Influences The Balance Of Healthy Gut Bacteria To Help Fight Infection At The Source
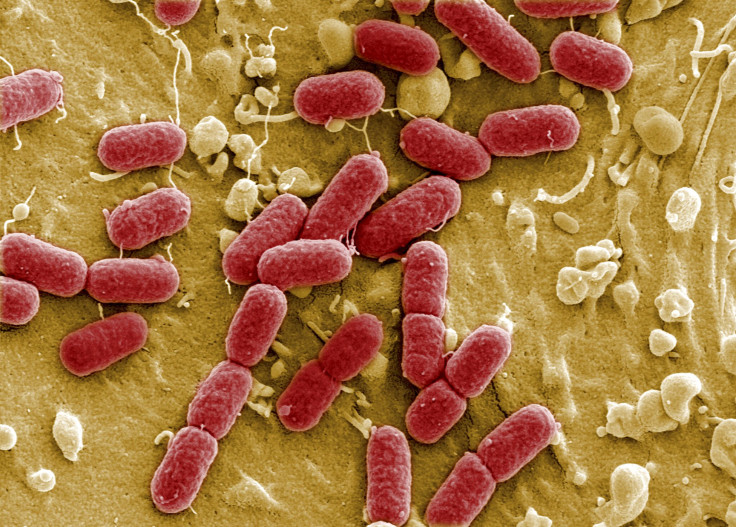
Homeostatis — the maintenance of a constant internal environment — is crucial to health, yet each of us has trillions of bacteria living inside our digestive tracts. How does this swarming colony of bacteria maintain homeostasis? Our immune system may be instrumental to this balance, say University of Chicago researchers, who discovered that a single binding protein on white blood cells influences whether or not mice produced a balanced gut microbiota.
The trillions of microbiota in our guts make an invaluable contribution to our metabolism by helping to break down complex carbohydrates and starches. Remarkably, they produce vitamins and hormones that direct the storage of fats. Many scientists believe our gut microbiota, which weigh up to 4 and a quarter pounds and collectively contain 3.3 million genes — a hundred times the number of human genes — act as an organ in-and-of themselves.
Over millions of years, the human body and its gut bacteria have evolved to live in harmony, to live symbiotically, according to Dr. Yang-Xin Fu, a professor in the University of Chicago Department of Pathology and senior author of the current study.
“This mutually beneficial relationship provides us with the ability to properly receive all of the nutrients from our food, and… the ability to limit harmful bacterial infections,” he stated in a press release.
For the current study, Fu and his colleagues investigated a specific intestinal immune cell, known as type 3 innate lymphoid cells (ILC3s). Upon examination, the researchers found ILC3s are less able to respond to harmful bacterial infections when they lack a protein called Id2.
The cells that lacked Id2 could not produce a specific molecule known to stimulate other cells in the intestines to produce antimicrobial peptides. Though these peptides helped to destroy bad bacteria entering the gut, normal bacteria entering or residing there are unharmed by antimicrobial peptides.
The Experiment
Conducting a little test, the team transferred microbiota from a mouse with dysfunctional ILC3s into germ-free mice — animals that had been bioengineered to lack bacteria in their guts. Separately, they transferred microbiota from a mouse with healthy, functional ILC3s into another set of germ-free mice. Next, they exposed both groups of mice to harmful bacteria. What did they find?
When exposed, the mice that had received healthy microbiota could fight off bacterial infection, while the mice that had received faulty ILC3s were highly susceptible to infection. This experiment, then, reveals how the immune system shapes our gut microbiota, acting as a guardian that maintains proper balance.
“Given the rapid rise of harmful bacteria that are resistant to antibiotics, it is paramount that scientists find methods of limiting harmful bacterial infections without the use of antibiotics,” Fu said. He hypothesizes that, in the future, infected patients might be helped by promoting “the development of good gut microbiota to indirectly kill harmful bacteria, instead of using antibiotics.”
Source: Guo X, Liang Y, Zhang Y, et al. Innate Lymphoid Cells Control Early Colonization Resistance against Intestinal Pathogens through ID2-Dependent Regulation of the Microbiota. Immunity. 2015.
Published by Medicaldaily.com



























