New Injectable Device May Revolutionize Parkinson's Disease Treatment: A Brief History Of Brain Probing

Long gone are the days of high-risk, invasive brain probing, when the brain was a mysterious arena we could barely touch without serious repercussions. While it’s still the most complicated organ, a research team from Harvard University has developed an electronic device that is so tiny and flexible it can be directly injected into the brain, where it can monitor brain activity and possibly even treated diseases like Parkinson’s or paralysis.
The researchers believe that this device is revolutionary, noting that it can do a lot inside the body without harming tissue or causing any complications — proven at least in mice, for now.
“We can precisely deliver these ultra-flexible electronics through a common syringe injection into virtually any kind of 3D soft material,” Charles Lieber, lead author of the study and a nanoscientist and nanotechnologist at Harvard University, told LiveScience. “The injection process and ultraflexible electronics introduce no damage to the targeted structures.”
To get it inside a mouse’s body, the researchers first create scaffolds as small flat sheets that consist of metal electrodes and silicone wires. Then, they attach sensors onto these meshes of metal and silicone that are about 90 percent empty space. These meshes roll up into a scroll when placed in liquid, then they can be injected through tiny syringes. Once inside the body, they revert back to their original, flat shape to do their thing.
Injecting these into the brains of mice during their experiments was promising, as it caused little to no bleeding and it recorded brain activity well. In addition, there’s no scar tissue or immune system response after the injection — even months afterwards. This is where this new device might revolutionize the brain probing scene, which has largely been plagued with such complications after implantations. Ultimately, if these meshes prove successful in the body for a long period of time, researchers could find ways to stimulate brain activity to treat Parkinson’s disease or other disorders.
“This opens up a completely new frontier where we can explore the interface between electronic structures and biology,” Lieber said in the Harvard press release. “For the past 30 years, people have made incremental improvements in micro-fabrication techniques that have allowed us to make rigid probes smaller and smaller.”
A Brief History Of Brain Probes

Prehistoric. Indeed, starting out, humans didn't have many options in studying the brain. Ancient trepanation is possibly the crudest and most primitive form of “brain-probing." It is actually considered the oldest surgical procedure that has archaeological evidence. Historically, trepanation, which involves drilling a hole into the skull, was used to let out ill demons from a person who was behaving abnormally. Later on, people used trepanation to treat migraines, seizures, and mental disorders — though it is largely considered a pseudoscience today.

1800s. Following years of research on animals using electroencephalography (EEG), psychiatrist Hans Berger recorded the first human EEG in 1924. While the technique isn’t invasive and isn’t used to treat diseases, it’s a diagnostic tool that involves covering the patient’s scalp with electrodes which record and monitor brain activity. EEGs are used to identify seizures and other neurological issues.
In the 1950s, Dr. Jose Delgado created the Stimoceiver, a device that he tested in the brain of a bull and made it charge and change direction. The Stimoceiver was a departure from earlier types of brain stimulators, which involved implanted electrodes in the brain that were connected to bulky equipment that recorded brain activity. This equipment was known to cause infections in patients and also limit movement.
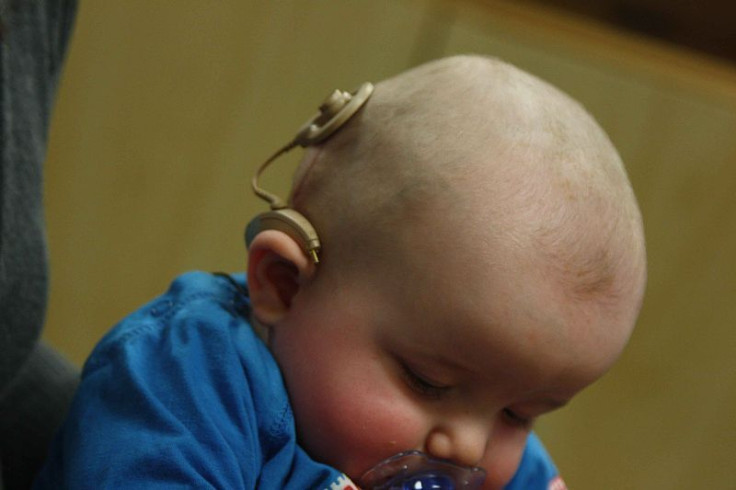
1970s. Cochlear implants, now in use by over 200,000 deaf people, assist patients in hearing by changing sound into electrical signals that are sent to the auditory nerve. While it's not directly inserted into the brain, the implant is placed into the ear and connected to an electrode on the brain, which stimulates hearing.
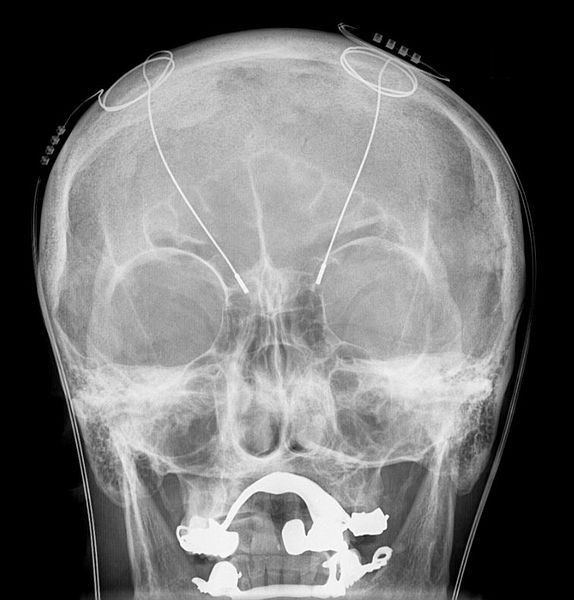
In 1997, the FDA approved deep brain stimulation to treat essential tremor. Later on, it was approved to treat Parkinson’s disease, dystonia, and even obsessive-compulsive disorder. During deep brain stimulation, which has been used to treat Parkinson’s disease and mental illness fairly successfully, a neurosurgeon inserts an electrode through a small opening in the skull and implants it in the brain. Then, electrical impulses are sent through the wire into the brain, which block abnormal electrical signals associated with Parkinson’s disease. This is still an invasive procedure, however, and thus not completely ideal.
In comparison to the potential of Lieber’s new device, these past examples of brain probing and monitoring seem outdated. With more research and clinical trials in humans, Lieber believes it will be transformative.
“Existing techniques are crude relative to the way the brain is wired,” Lieber said in the press release. “Whether it’s a silicon probe or flexible polymers … they cause inflammation in the tissue that requires periodically changing the position or the stimulation. But with our injectable electronics, it’s as if it’s not there at all. They are one million times more flexible than any state-of-the-art flexible electronics and have subcellular feature sizes. They’re what I call ‘neuro-philic’ — they actually like to interact with neurons.”
Source: Liu J, Fu T, Cheng Z, Hong G, Zhou T, Lieber C. Syringe-injectable electronics. Nature Nanotechnology. 2015.