Promising HIV Treatments: What Scientists Are Working On Right Now


This question originally appeared on Quora. Answer by Christopher VanLang, PhD.
A challenging question since scientists do have a potential path forwards to cure HIV infection, but they don’t make any practical sense since physicians shouldn’t resort to aggressive cures for HIV when the current anti-retroviral therapies (ART) are far less risky.
What we should be doing is focusing on small molecules and vaccines that allow us to eliminate latent HIV infection.
Based on several case studies, one could essentially cure HIV if they:
- Perform bone marrow transplants with a CCR5 deficient stem cell transplant similar to what was done for the Berlin patient.
- Use gene therapy to knockout key pathways in T-Cells namely CCR5.
- Use gene therapy to knockout key pathways in HIV.
- Add decoy cells to extract HIV from latency.
However, we would never realistically pursue any of these mechanisms of action in the near future unless an emergency requires it (like a bone marrow transplant to treat leukemia). The risks and costs of the above options outweigh the benefits compared to taking pills. While CRISPR/Cas9 is the new hot technology, it doesn’t make medical sense to apply a non-specific excision system using a Lentiviral vector in cells that will clear out in a couple of months thus requiring a new bolus of Lentiviral vectors unless you figure a way to target latently infected T-cells. Furthermore, these therapeutic strategies could not be successfully applied in third world countries where HIV is a more serious problem. We should look to more promising alternatives.
There are numerous challenges with developing a non-gene editing option to treat HIV. While patients are now effectively cured using ART using a combination of nucleoside analog reverse transcriptase inhibitors (NRTIs) and protease inhibitors, these treatments are effective in preventing further effects of HIV but do not remove the presence of HIV from the body. HIV is particularly difficult to remove because the virus can stay latent in the CD4+ T-cell genome for decades, and the constant threat of the reactivation of these latent HIV reservoirs require patients to strictly adhere to their ART regimens. Furthermore, drug resistant variants can evolve while in the latent phase. Despite several efforts of increased dosing with ART along with the introduction of anti-integrase drugs like raltegravir to prevent the reintroduction of HIV into the genome, the levels of latent HIV have not been affected indicating that the HIV is stably integrated into the resting memory CD4+ T-cells.
To enable long-term resistance of latent HIV reactivation without a life-time dependency on drugs, we should be focusing on two areas:
- Activation of HIV gene expression from resting CD4+ T-cells to reduce latent HIV
- Generation of broadly neutralizing antibodies using an anti-HIV vaccine.
Reduce latent HIV
HIV resides in resting memory CD4+ T-cells which continuously regenerate and result in the life-long infection of HIV. Fortunately, there is a model Jurkat T-Cell line, J-Lat, which contains latent HIV. This model cell line can be used to further studies of the mechanisms of reactivation. Humanized mouse models and the SIV-macaque model are also useful models of latent HIV infection.
There are several remaining technical challenges to pursuing this path. First, it is still unclear how HIV establishes latent infection of these resting CD4+ T-cells since they have relatively low expressions of CCR5. Secondly, because of the difficulty of selectively activating latently infected cells from the HIV-free T-cells, global activation of resting T-cells using anti-CD3 antibodies or T-cell activation proteins like Interleukin-7 and Interleukin-2 have lead to severe adverse effects from cytokine release. Only 1 in 1 million resting T-cells are infected.
However, several studies on the epigenetics of HIV suppression has identified several pathways that suppress HIV activation.
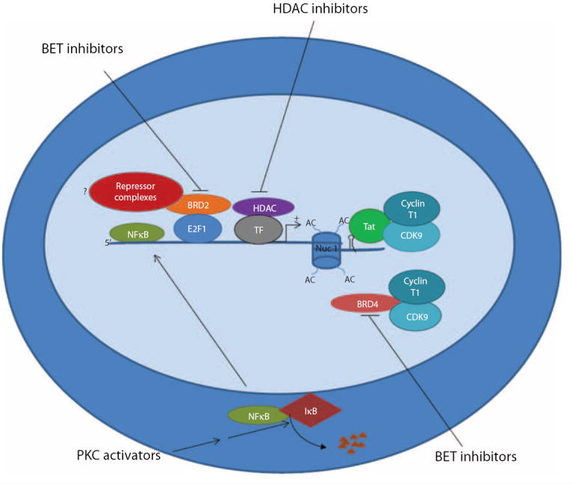
As such, groups have deferred to small molecule activators like bryostatin which is hypothesized to activate CD4. Certain clinical trials are exploring the use of the HDAC inhibitor vorinostat along with ART to activate HIV and transcriptional studies showed a significant change in viral RNA expression. At the moment, future clinical trials using vorinostat are mainly being investigated in high risk patients with HIV infection and cancer and this should serve as a strong proof of concept trial to test the effect of using HDAC inhibitors.
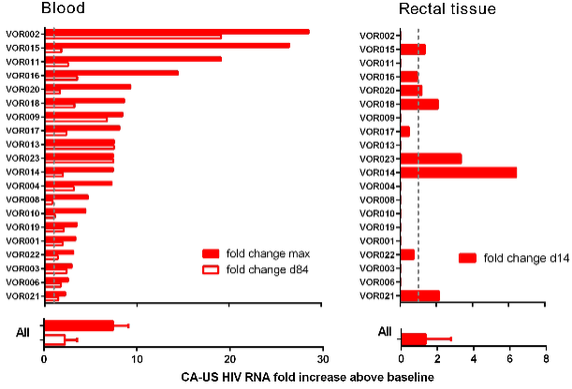
More likely, to see further use of these latent HIV T-Cell activators , we would need to perform cell-based screens that select for compounds that work only on infected cells and not on healthy cells. Until we have a good understanding of the risks, we wouldn’t be able to make sufficient progress in treating patients overseas. However, early Phase I work has suggested that we have the ability to drive out HIV from latency.
Anti-HIV vaccine
A huge amount of work has been put into the development of broadly neutralizing antibodies and the mechanisms to generate an immune response to both a preventative and therapeutic vaccine that would infer immunity to latent HIV.
The challenges behind HIV vaccine research is well documented on Quora and I highly recommend looking at Brian Dickerson's answer to Why has developing an effective vaccine for HIV proven so difficult? Some of the major key points is that the exposed domain ENV of the HIV virus are rapidly evolving and are protected by several glycans which cover >70% of the protein.
A huge focus has been place on neutralizing antibodies to GP120, the CD4 binding site of HIV. Alternatively, one could develop broadly neutralizing antibodies towards GP41 which is better conserved. The RV144 “Thai trial” using the prime-boost HIV vaccine was determined to be largely ineffective despite successfully generating an immune response.
As outlined in [a 2013 study,] Broadly neutralizing antibodies and the search for an HIV-1 vaccine: the end of the beginning, a huge number of the broadly neutralizing antibodies are directed towards the GP120 end of the ENV protein (the bottom). However, there are a few antibodies that will target the membrane-proximal external region (MPER) of GP41 notably the 10e8 antibody and the 35O22 antibody.
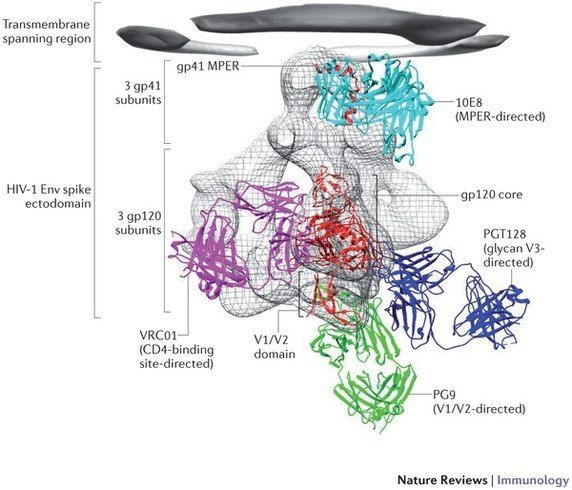
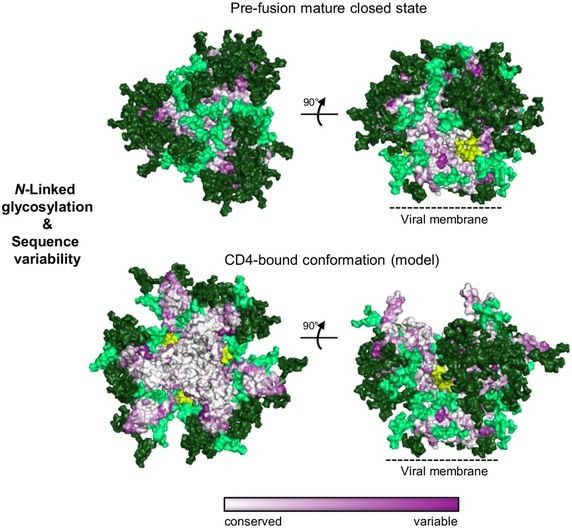
If you look at the sequence conservation of the ENV protein and the location of the glycans (shown in green), you would notice that the most heterogeneous sites and glycosylated sites are on the GP120 side of the protein rather than the GP41 end. Note that the highly conserved sites of the ENV protein are exposed only during CD4 binding.
The role of GP41 has been long recognized by Peter Kim (former President of Merck Research Labs). In a string-loaded mechanism, GP41 forms a coiled-coil that stacks the N-heptad repeat (NHR) on the C-heptad repeat (CHR). This domain in turn recognzies CCR5 and forces a pre-fusion complex that enable viral fusion. This is a similar mechanism to influenza and Ebola.
Along with these vaccine efforts, there are a lot of promising drug discovery efforts to develop small molecule and peptide inhibitors of viral fusion. Fusion inhibitors will bind to the conserved region of GP41 in two mechanisms, the C-peptide and the D-peptide. Essentially, a mimic of the CHR but making a synthetic C-peptide can act as a transient fusion intermediate and block the interaction of GP41 with CCR5. Alternatively, a hydrophobic pocket that can be targeted by small molecule inhibitors is known and can be blocked by D-peptides. Enfuvirtide is the only currently approved GP41 inhibitor working as a C-peptide but is fairly limited as an HIV-1 specific drug with a very short half-life and other pharmacological limits due to its peptide mechanism. The particular challenge of these efforts is the general difficulty of developing what amounts to a protein-protein small molecule inhibitor.
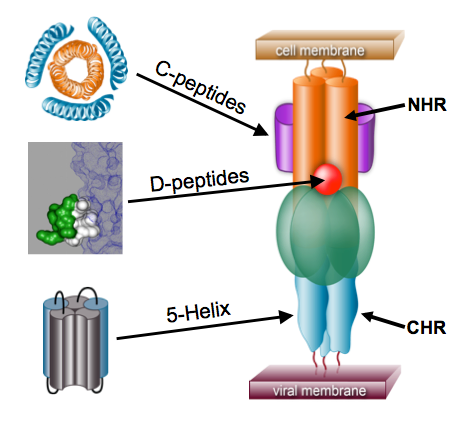
The holy grail would be generate an immune response that creates a broadly neutralizing antibody against this highly conserved pre-hairpin intermediate. The challenge of course is the difficulties of stabilizing this intermediate for the purpose of generating an immune response. However, a peptide-based antigen would provide a more reproducible immune response compared to HIV based vaccines and prime/boost vaccines which are more likely to develop a GP120 immune response. Furthermore, an immune response against the N-heptad peptide of GP41 would serve as both a preventative and therapeutic vaccine
In summary, I propose two potential mechanism of action for HIV treatment that should be scaleable and adoptable for use in third world countries.
The first is the use of activators of HIV expression via a combination of HDAC and PKC inbitors may drag out the hidden HIV and expose it to antiviral therapies that will kill and remove the virus.
The second is the development of a stabilized pre-hairpin intermediate of GP41 as a potential vaccine target.
More from Quora:
Published by Medicaldaily.com



























