What Are Monoclonal Antibodies? New Immunotherapy Melts Skin Cancer Tumor
Monoclonal antibodies are a type of drug classified as "immunotherapy" due to the fact they guide your immune system to recognize and then attack dangerous pathogens (such as cancer). Last week the New England Journal of Medicine reported how two monoclonal antibodies were so effective against skin cancer, they melted away one woman’s tumor and left behind an empty space after just one treatment. Maybe this isn't the optimal response, still this profound result speaks to the effectiveness of this relatively recent category of drugs, which the Food and Drug Administration first began to approve as cancer treatments about 20 years ago.
So how exactly do these immunotherapies work? To understand this, it is important to acknowledge cancer’s most powerful characteristic — it’s ability to deceive the immune system.
Similar to terrorist cells living and working in plain sight, cancer cells live and multiply within your body without ever being recognized as dangerous. Again and again, your immune system misreads the chatter of these cells, and so allows them to multiply and grow. This is the exact problem scientists created immunotherapies to address.
Monoclonal antibodies work by essentially drawing a target on cancer cells.
Once the antigen within this deadly invader has been identified, immune system antibodies will attach to it and then destroyer cells within your body’s defense system will demolish it.
Monoclonal antibodies, then, are engineered in a lab — repeatedly cloned, actually, from a specially designed antibody — to attach to a specific antigen on the surface of cancer cells. In this way the monoclonal antibody marks the cancer cell, making it more visible to the immune system.
For the study, then, researchers investigated the combined use of two FDA-approved monoclonal antibodies (sometimes called MAbs — hence the names of these drugs both end in "mab") in 142 patients with metastatic melanoma. Some of the patients received ipilimumab (Yervoy) combined with nivolumab (Opdivo) and then the researchers compared their results to that of patients receiving doses of ipilimumab alone. (Both of these immunotherapy drugs are produced by Bristol-Myers Squibb.) Though these immunotherapy drugs work in different ways, both inspire the activation and proliferation of T-cells, cancer-fighting immune cells.
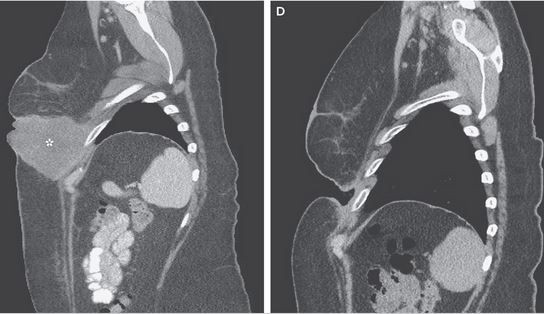
The combined efforts of the two monoclonal therapies worked very well for most patients and exceptionally well for one… so well, in fact, the authors felt the need to warn other doctors about the possibility of a robust response. The one patient, a 49-year-old woman whose medical history included the persistent return of tumors removed by surgery, “had a rapid eradication of a large tumor mass after a single treatment with combination immunotherapy,” wrote the authors. In fact, a gaping hole was left behind.
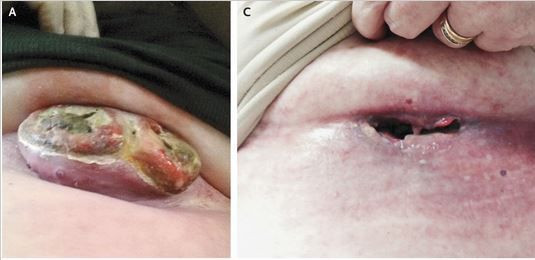
“It is ironic that we are now concerned about the possibility of overly vigorous antimelanoma responses,” the authors concluded. Medicine has tempered other cures, adjusting them to both the patient and the disease. With hope, the same will be done with immunotherapies.
Source: Postow MA, Chesney J, Pavlick AC, et al. Nivolumab and Ipilimumab versus Ipilimumab in Untreated Melanoma. NEJM. 2015.
Published by Medicaldaily.com



























