The Price Of Science: OHRP Identifies Infant Study Where Parents Were Misled Regarding Health Risks Of Test Treatment
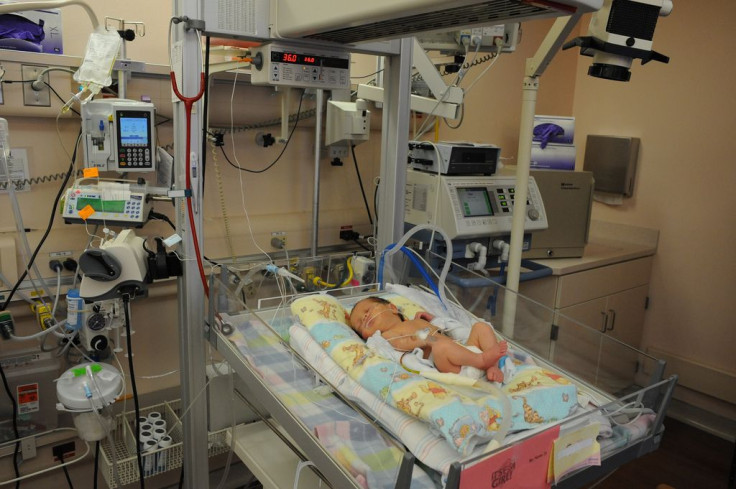
While the completion of scientific research projects relies on participants who are willing to take risks to benefit others. While study participants usually consent to these studies and are often compensated, what happens when those studied are too young to consent?
A recent National Institutes of Health (NIH) funded study called SUPPORT which followed 1,316 premature babies. Their oxygen levels were maintained at either too high or too low saturation levels immediately after their birth to study which conditions would lead to positive or adverse effects, in order to help premature babies develop properly.
Depriving babies, especially those born prematurely, of oxygen is highly dangerous. Often, prematurely born babies have poorly developed lungs. This means they struggle to take up enough oxygen into their bodies. They usually develop chronic issues as a result of their premature birth. However, denying a prematurely born child oxygen for the sake of a study, without informing his or her parents of the serious health and developmental a lack of oxygen can cause has been called into question.
The American government's Office of Human Research Protections (OHRP), pursuant to a complaint about the SUPPORT study two years ago, concluded its investigation with a letter to the lead site in the study, the University of Alabama in Birmingham. The letter cited and criticized multiple instances of misinformation about risks in the consent forms parents signed, compared with all of the safety information delineating higher risk for the infants with low levels of oxygen in the published protocol of the study. The study's protocol was clear about wanting to find out whether death or eye damage was more common as a result of the oxygen level the child received.
The protocol admits that the higher oxygen levels were safe or "more conventional", but no consent forms ever listed the lower oxygen level as "less conventional" or dangerous. Many of the consent forms misleadingly stated that because all of the treatments proposed in this study are "standard of care," there would be no expected increase in risk to the infants in either groups. Meanwhile, the oxygen levels could potentially kill the infants receiving less.
The weak defense of the SUPPORT research group was that their study intended to focus on the change in oxygen's effect on eye development, and that death rates were never their focus.
"They did not expect an increased rate of death in the low oxygen group, this clearly possible risk did not need to be disclosed in the consent forms. The fact that death was one of the primary stated outcomes of the study seems to have escaped them," Dr. Sidney M. Wolfe, founder and senior adviser at the Health Research Group at Public Citizen explains.
"To seek truly Informed consent for participation in a research study is to oblige parents to listen to complex medical arguments that spell out the uncertainties of current practice," states neonatologist Dr. Neena Modi states in an editoria. "Although it might be argued that parents have a right to know about all aspects of their baby's care, this would mean that, in many instances, distressed parents were forced to make decisions that they would not normally be asked to make."
Modi admits that while studies like these are needed to improve care in circumstances once thought to be hopeless, parents must be given all the information necessary to make the right decision, instead of feeling trapped into a study as a result of their unfortunate circumstance.
Some argue that participants who are kept overly informed of treatments tested may become biased, misreport symptoms, and ruin the study. However, in treatments that can ultimately cause death, such as the kind tested on the premature infants, consent must start with full disclosure of any and all risks.
While research to improve care and medicines is important, so is preserving the safety and lives of participants. More must be done to prevent misleading potential participants and ensure they are fully informed of the risks they are taking on.
Source: Wolfe SM. US Study Criticized for Experimentation with Premature Infants. BMJ. 2013.



























